Stability Enhancement for All Inorganic CsPbX3 Perovskite Solar Cells via Metal Ions Doping
-
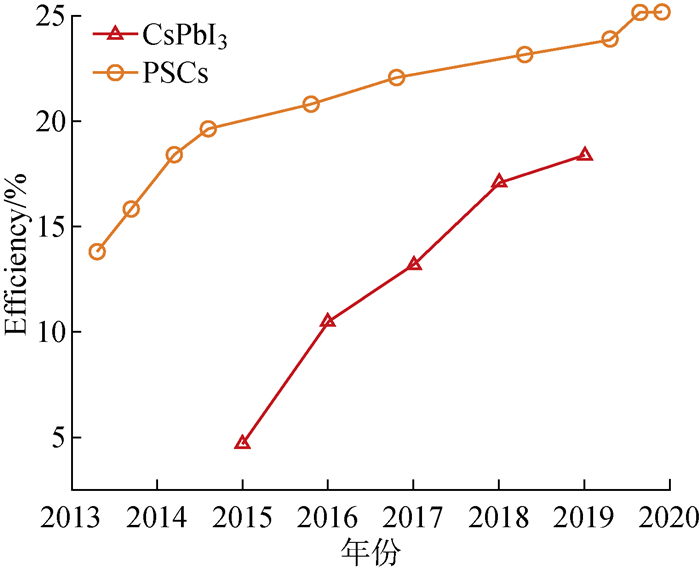
摘要: 钙钛矿太阳能电池具有光电转换效率高、成本低等优势,有望成为新一代光伏技术。然而,钙钛矿材料本身较低的稳定性限制了其商业化应用。因此,提高钙钛矿材料的稳定性对进一步推进钙钛矿太阳能电池的实用化至关重要。综述了金属离子掺杂在全无机钙钛矿太阳能电池的结构、组成及稳定性等方面的进展,重点介绍了以A位和B位金属离子2类典型的离子掺杂稳定全无机钙钛矿太阳能电池的研究工作。最后,提出了金属离子掺杂作为全无机钙钛矿太阳能电池稳定性强化策略所面临的机遇和挑战。Abstract: Perovskite solar cells (PSCs) are expected to be the new generation of photovoltaic technologies, owning to their high photoelectric conversion efficiency and low cost. However, the lower stability of perovskite materials restricts their commercial application. Therefore, it is vital to improve the stability of perovskite materials to further promote the practicality of perovskite solar cells. The progress of metal ion doping strategy in tuning the structure, composition, and stability of all-inorganic perovskite solar cells was reviewed, with emphasis on the metal ions doping at the A- and B-site of CsPbX3 perovskites. Finally, the opportunities and challenges of metal ion doping to develop highly efficient and stable all-inorganic perovskites solar cells were put forward.
-
Key words:
- solar cells /
- all inorganic perovskite /
- metal ion doping /
- stability
-
图 4 (a) CsPbI2Br钙钛矿器件结构以及Mn2+离子掺杂的示意图[28];(b) Zn(蓝色球)在钙钛矿晶格间隙的示意图[32];(c) Eu3+离子掺杂CsPbI3钙钛矿的结构示意图[34];(d)Cu2+离子掺杂CsPbI2Br钙钛矿太阳能电池器件的J-V曲线[40]
Figure 4. (a) Schematic structure of the device and illustration of the Mn2+ ions doping modes: interstitial and substituting[28], (b) The scheme of interstitial Zn (blue ball) in perovskite lattice[32], (c) Schematic structure of the Eu3+ ions doping CsPbI3 perovskite[34], (d) J-V curves of Cu2+ ions doping CsPbI2Br perovskite solar cells[40]
表 1 全无机钙钛矿中的掺杂离子及其半径和对应的功率转换效率
Table 1. Doped ions in all-inorganic perovskites, their radius and corresponding PCE
掺杂离子 离子半径/pm 钙钛矿组分 效率/% 参考文献 Mn2+ 67 CsPbI2Br 13.47 [28] Sr2+ 113 CsPbI2Br 11.3 [29] Ca2+ 100 CsPbI3 13.3 [30] Ba2+ 135 CsPbI2Br 14.0 [31] Zn2+ 74 CsPbI2Br 13.6 [32] Sn2+ 112 CsPbI2Br 11.33 [33] Eu3+ 95 CsPbI3 6.0 [34] Eu2+ 117 CsPbI2Br 13.71 [35] Bi3+ 108 CsPbI3 13.21 [36] Ni2+ 69 CsPbI2Br 13.88 [37] Ge2+ 73 CsPbI2Br 10.8 [38] Nb5+ 64 CsPbI2Br 10.42 [39] Cu2+ 73 CsPbI2Br 16.15 [40] -
[1] LIANG Z, SU M, WANG H, et al. Characteristics of a silicon nanowires/PEDOT: PSS heterojunction and its effect on the solar cell performance[J]. ACS Applied Materials & Interface, 2015, 7(10): 5830-5836. [2] CHANG C H, LEE Y L. Chemical bath deposition of CdS quantum dots onto mesoscopic TiO2 films for application in quantum-dot-sensitized solar cells[J]. Applied Physics Letters, 2007, 91(5): 053503. doi: 10.1063/1.2768311 [3] WU Y Z, ZHU W H. Organic sensitizers from D-π-A to D-A-π-A: Effect of the internal electron-withdrawing units on molecular absorption, energy levels and photovoltaic performances[J]. Chemical Society Reviews, 2013, 42(5): 2039-2058. doi: 10.1039/C2CS35346F [4] DIAO Y, ZHOU Y, MANNSFLED S C B, et al. Flow-enhanced solution printing of all-polymer solar cells[J]. Nature Communications, 2015, 6: 7955. doi: 10.1038/ncomms8955 [5] HAO M M, BAI Y, CHENG H M, et al. Ligand-assisted cation-exchange engineering for high-efficiency colloidal Cs1-xFAxPbI3 quantum dot solar cells with reduced phase segregation[J]. Nature Energy, 2020, 5(1): 79-88. doi: 10.1038/s41560-019-0535-7 [6] JU D X, JIANG X M, YIN J, et al. Research progress of organic-inorganic composite perovskite single crystal materials[J]. Journal of Technology, 2019, 19(2): 95-109. http://d.old.wanfangdata.com.cn/Periodical/shyyjsxyxb201902001 [7] CHEN W, WU Y Z, GRATZEL M, et al. Efficient and stable large-area perovskite solar cells with inorganic charge extraction layers[J]. Science, 2015, 350(6263): 944-948. doi: 10.1126/science.aad1015 [8] ZHANG F, ZHONG H Z, CHEN C, et al. Brightly luminescent and color-tunable colloidal CH3NH3PbX3 (X = Br, I, Cl) quantum dots: Potential alternatives for display technology[J]. ACS NANO, 2015, 9(4): 4533-4542. doi: 10.1021/acsnano.5b01154 [9] The National Renewable Energy Laboratory. Best Research-Cell Efficiency Chart[OL]. https://www.nrel.gov/pv/assets/pdfs/pv-efficiency-chart.[2020-03-20]. [10] NIU G D, LI W Z, WANG L D, et al. Study on the stability of CH3NH3PbI3 films and the effect of post-modification by aluminum oxide in all-solid-state hybrid solar cells[J]. Journl of Materials Chemistry A, 2014, 2(3): 705-710. [11] BERHE T A, SU W N, CHEN C H, et al. Organometal halide perovskite solar cells: Degradation and stability[J]. Energy & Environmental Science, 2016, 9(2): 323-356. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ0232887037/ [12] EPERON G E, STRANKS S D, MENELAOU C, et al. Formamidinium lead trihalide: A broadly tunable perovskite for efficient planar heterojunction solar cells[J]. Energy & Environmental Science, 2014, 7(3): 982-988. [13] POLMAN A, KNIGHT M, GARNNET E C, et al. Photovoltaic materials: Present efficiencies and future challenges[J]. Science, 2016, 352(6283): 307. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ0231721918/ [14] RIPOLLES T S, NISHINAKA K, OGOMI Y, et al. Efficiency enhancement by changing perovskite crystal phase and adding a charge extraction interlayer in organic amine free perovskite solar cells based on cesium[J]. Solar Energy Materials and Solar Cells, 2016, 144: 532-536. doi: 10.1016/j.solmat.2015.09.041 [15] FROLOVA L A, ANOKHIN D V, TROSHIN P A, et al. Highly efficient all-inorganic planar heterojunction perovskite solar cells produced by thermal coevaporation of CsI and PbI2[J]. The Journal of Physical Chemistry Letters, 2017, 8(1): 67-72. doi: 10.1021/acs.jpclett.6b02594 [16] WANG Y, ZHANG T Y, KAN M, et al. Bifunctional stabilization of all-inorganic α-CsPbI3 perovskite for 17% efficiency photovoltaics[J]. Journl of American Chemistry Society, 2018, 140(39): 12345-12348. doi: 10.1021/jacs.8b07927 [17] WANG P Y, ZHANG X W, YANG X L, et al. Solvent-controlled growth of inorganic perovskite films in dry environment for efficient and stable solar cells[J]. Nature Communications, 2018, 9: 2225. doi: 10.1038/s41467-018-04636-4 [18] WANG Y, DAR M I, QI Y B, et al. Thermodynamically stabilized β-CsPbI3-based perovskite solar cells with efficiencies >18%[J]. Science, 2019, 365(6453): 591-595. doi: 10.1126/science.aav8680 [19] MARRONNIER A, ROMA G, BOYER-RICHARD S, et al. Anharmonicity and disorder in the black phases of cesium lead iodide used for stable inorganic perovskite solar cells[J]. ACS Nano, 2018, 12(4): 3477-3486. doi: 10.1021/acsnano.8b00267 [20] JULIAN A. STEELE J A, JIN H D, et al. Thermal unequilibrium of strained black CsPbI3 thin films[J]. Science, 2019, 365(6454): 679-684. doi: 10.1126/science.aax3878 [21] WANG Q, ZHENG X, DENG Y, et al. Stabilizing the α-phase of CsPbI3 perovskite by sulfobetaine zwitterions in one-step spin-coating films[J]. Joule, 2017, 1(2): 371-382. doi: 10.1016/j.joule.2017.07.017 [22] WANG K, JIN Z W, LIANG L, et al. All-inorganic cesium lead iodide perovskite solar cells with stabilized efficiency beyond 15%[J]. Nature Communications, 2018, 9: 4544. doi: 10.1038/s41467-018-06915-6 [23] GREEN M A, HO-BAILLIE A. Perovskite solar cells: The birth of a new era in photovoltaics[J]. ACS Energy Letters, 2017, 2: 822-830. doi: 10.1021/acsenergylett.7b00137 [24] LI C, LU X, DING W, FENG L, et al. Formability of ABX3 (X= F, Cl, Br, I) halide perovskites[J]. Acta Crystallographica Section B Structural Science, 2008, 64: 702-707. doi: 10.1107/S0108768108032734 [25] NAM J K, KIM D, PARK J H, et al. Potassium incorporation for enhanced performance and stability of fully inorganic cesium lead halide perovskite solar cells[J]. Nano Letters, 2017, 17(3): 2028-2033. doi: 10.1021/acs.nanolett.7b00050 [26] XIANG S S, YANG S H, CHEN H N, et al. Natrium doping pushes the efficiency of carbon-based CsPbI3 perovskite solar cells to 10.7%[J]. Science, 2019, 15: 156-164. [27] GUO Y X, ZHAO F, TAO J H, et al. Efficient and hole transporting layer-free CsPbI2Br planar heterojunction perovskite solar cell via rubidium passivation[J]. ChemSusChem, 2019, 12(5): 983-989. doi: 10.1002/cssc.201802690 [28] BAI D L, ZHANG J R, JIN Z W, et al. Interstitial Mn2+-driven high-aspect-ratio grain growth for low-trap-density microcrystalline films for record efficiency CsPbI2Br solar cells[J]. ACS Energy Letters, 2018, 3(4): 970-978. doi: 10.1021/acsenergylett.8b00270 [29] LAU C F J, ZHANG M, HU L, et al. Strontium doped low temperature processed CsPbI2Br perovskite solar cells[J]. ACS Energy Letters, 2017, 2(10): 2319-2325. doi: 10.1021/acsenergylett.7b00751 [30] LAU C F J, DENG X, ZHENG J, et al. Enhanced performance via partial lead replacement with calcium for CsPbI3 perovskite solar cell exceeding 13% power conversion efficiency[J]. Journl of Material Chemistry A, 2018, 6(14): 5580-5586. doi: 10.1039/C7TA11154A [31] XIANG W, WANG Z, GRATZEL M, et al. Ba-induced phase segregation and band gap reduction in mixed-halide inorganic perovskite solar cells[J]. Nature Communications, 2019, 10: 4686. doi: 10.1038/s41467-019-12678-5 [32] YANG F, HIROTANI D, KAPIL G, et al. All-inorganic CsPb1-xGexI2Br perovskite with enhanced phase stability and photovoltaic performance[J]. Angewandte Chemie International Edition, 2018, 57(39): 12745-12749. doi: 10.1002/anie.201807270 [33] LIANG J, ZHAO P, WANG C, et al. CsPb0.9Sn0.1IBr2 based all-inorganic perovskite solar cells with exceptional efficiency and stability[J]. Journl of American Chemistry Society, 2017, 139(40): 14009-14012. doi: 10.1021/jacs.7b07949 [34] JENA A K, KULKARNI A, SANEHIRA Y, et al. Stabilization of α-CsPbI3 in ambient room temperature conditions by incorporating Eu into CsPbI3[J]. Chemistry Materials, 2018, 30(19): 6668-6674. doi: 10.1021/acs.chemmater.8b01808 [35] XIANG W, WANG Z, KUBICKI D J, et al. Europium-doped CsPbI2Br for stable and highly efficient inorganic perovskite solar cellss[J]. Joule, 2019, 3(1): 205-214. doi: 10.1016/j.joule.2018.10.008 [36] HU Y, BAI F, LIU X, et al. Bismuth incorporation stabilized α-CsPbI3 for fully inorganic perovskite solar cells[J]. ACS Energy Letters, 2017, 2(10): 2219-2227. doi: 10.1021/acsenergylett.7b00508 [37] CHEN L J, WAN L, LI X D, et al. Inverted all-inorganic CsPbI2Br perovskite solar cells with promoted efficiency and stability by nickel incorporation[J]. Chemistry of Materials, 2019, 31(21): 9032-9039. doi: 10.1021/acs.chemmater.9b03277 [38] SUN H R, ZHANG J, SHANG M H, et al. Pb-reduced CsPb0.9Zn0.1I2Br thin films for efficient perovskite solar cells[J]. ACS Energy Letters, 2019, 9(25): 1900896. [39] GUO Z L, ZHAO S, HAYASE S, et al. Niobium incorporation into CsPbI2Br for stable and efficient all-inorganic perovskite solar cells[J]. ACS Applied Materials & Interfaces, 2019, 11(22): 19994-20003. [40] WANG K L, WANG R, WANG Z K, et al. Tailored phase transformation of CsPbI2Br films by copper(Ⅱ) bromide for high-performance all-inorganic perovskite solar cells[J]. Nano Letters, 2019, 19(8): 5176-5184. doi: 10.1021/acs.nanolett.9b01553 -





 下载:
下载:







 地址:上海市漕宝路120号
地址:上海市漕宝路120号 电话:021-64691562
电话:021-64691562  邮箱:
邮箱: 版权所有:《应用技术学报》编辑部
版权所有:《应用技术学报》编辑部